Monday, August 26th
Objective:
Students will demonstrate their current knowledge of science
Assignments:
School City - Use the bookmark under "Granite SD" to find School City. Your log in is your Student ID# (9 #) and your regular log-in password. When completed...
EdPuzzle - we will use EdPuzzle for homework this semester. Go to www.edpuzzle.com and click on "Sign Up"
Students will demonstrate their current knowledge of science
Assignments:
- School City Pre-Test Science 8
- EdPuzzle.com - Homework #1
School City - Use the bookmark under "Granite SD" to find School City. Your log in is your Student ID# (9 #) and your regular log-in password. When completed...
EdPuzzle - we will use EdPuzzle for homework this semester. Go to www.edpuzzle.com and click on "Sign Up"
Tuesday, 27 August
OBJECTIVE: Students will learn what tasks are involved in doing a lab
ASSIGNMENTS: Students will complete the observations, group data table, conclusion, and graph for the Bubble Lab we did last Friday. All students, regardless of whether you blew the bubbles or not are required to complete these sections.
ASSIGNMENTS: Students will complete the observations, group data table, conclusion, and graph for the Bubble Lab we did last Friday. All students, regardless of whether you blew the bubbles or not are required to complete these sections.
| bubble.docx | |
| File Size: | 16 kb |
| File Type: | docx |
Wednesday, 28 August
OBJECTIVE: Students will learn how to start a phenomenon.
ASSIGNMENTS:
ASSIGNMENTS:
- Initial Answer in Science Journal
- Team Initial Answer Poster and Presentation
Are all atoms the same?
INITIAL ANSWER - REQUIRED
OBJECTIVE: Students will share with team members what they know about atoms and come to a team consensus (team answer)
PURPOSE: To think about what you already know about an atom, and to start think about what you don't know about the atom, yet.
STANDARD: 8.1.1: Develop a model to describe the scale and proportion of atoms and molecules. Emphasize developing atomic models of elements and their numbers of protons, neutrons, and electrons, as well as models of simple molecules.
INSTRUCTIONS:
OBJECTIVE: Students will share with team members what they know about atoms and come to a team consensus (team answer)
PURPOSE: To think about what you already know about an atom, and to start think about what you don't know about the atom, yet.
STANDARD: 8.1.1: Develop a model to describe the scale and proportion of atoms and molecules. Emphasize developing atomic models of elements and their numbers of protons, neutrons, and electrons, as well as models of simple molecules.
INSTRUCTIONS:
- In your science journal, Write the heading "Are All Atoms the Same?" on the next left hand page of your science journal - DON'T FOR GET TO PUT THIS HEADING IN YOUR TABLE OF CONTENTS
- Under the title, write Initial Answer. Then, write your individual initial answer - this should include what you already know about Atoms
- Share your answers with your team
- Team combines individual answers to develop a team answer
- Team answer is depicted in poster. Everyone should have a different colored marker and contribute to the creating the poster. More pictures and less words are ideal for this poster
- The Team Reporter then presents the poster to class
LEARNING OBJECTIVE: Students will brainstorm questions that will help lead them to the answer for our Phenomenon Question: "Are all Atoms the Same?"
PURPOSE: To understand the concepts that will enable us to answer the phenomenon question, "Are Atoms all the same?"
STANDARD: 8.1.1: Develop a model to describe the scale and proportion of atoms and molecules. Emphasize developing atomic models of elements and their numbers of protons, neutrons, and electrons, as well as models of simple molecules.
PURPOSE: To understand the concepts that will enable us to answer the phenomenon question, "Are Atoms all the same?"
STANDARD: 8.1.1: Develop a model to describe the scale and proportion of atoms and molecules. Emphasize developing atomic models of elements and their numbers of protons, neutrons, and electrons, as well as models of simple molecules.
INSTRUCTIONS:
1. On the right side of your next journal page: Come up with 5 questions that you think will help you find the answer to our phenomenon question, "Are Atoms all the same?" Write these questions in your science journal. You will be answering these questions too, so leave room to write your answers.
1. On the right side of your next journal page: Come up with 5 questions that you think will help you find the answer to our phenomenon question, "Are Atoms all the same?" Write these questions in your science journal. You will be answering these questions too, so leave room to write your answers.
3. Use the resources below to help you do your research for your questions/answers as well as your MINDMAP - use at least 2 of the sources below:
CK-12
8th Grade Online Textbook - pages 13 - 15
Kids Discover Online - register for our class and then explore the two articles on atoms
Chem4Kids
Ducksters
Wikipedia
CK-12
8th Grade Online Textbook - pages 13 - 15
Kids Discover Online - register for our class and then explore the two articles on atoms
Chem4Kids
Ducksters
Wikipedia
THURSDAY, 29 August
Friday, 30 August
OBJECTIVES:
- Students will learn about the size and history of the atom
- Atom Size Activity
- Cornell Notes for Videos
Who came up with the idea of an atom?
HOW BIG IS AN ATOM? - REQUIRED
Team Work
Team Work
OBJECTIVE: Students will learn about the initial ideas of an atom.
PURPOSE: To see how small an atom really is
STANDARD: 8.1.1 : Develop a model to describe the scale and proportion of atoms and molecules. Emphasize developing atomic models of elements and their numbers of protons, neutrons, and electrons, as well as models of simple molecules.
Bill Nye Cuts Cheese
Activity
INSTRUCTIONS:
1. Decide on Team Roles
2. Facilitator, read the Team Norms to your team
3. Resource Manager get the needed materials for this lab from the demo table and hand them out to your team members.
4. Team Captain will read the directions.
Directions for Activity:
A. Title the next left page in your science journal "How big is an atom?" Record the title in your table of contents along with the page number.
B. Glue the red piece of paper on this science journal page
C. Write down in your science journal, a prediction on how many times you think you will be able to cut the white piece of paper in half. You will be cutting the paper until you cannot cut a piece in half.
D. Cut the white sheet of paper in half.
E. Take one half of this paper and put it to the side. Cut the other one in half again. (Make all cuts perpendicular to the first.)
F. Repeat this process, until you can no longer cut the paper in half.
G. Record the number of cuts you were able to make in your science journal.
H. Guess how many cuts it would take to obtain a piece of paper as small as an atom. Write your guess in your science journal.
I. Together as a team, read the the document entitled "What is an Atom?"
J. Follow the directions for Experiment 2
K. Answer all of the questions from "What is an Atom?" in your science journal. Work through all questions together as a team. Discuss the answers together before deciding on an answer to write in your science journal.
PURPOSE: To see how small an atom really is
STANDARD: 8.1.1 : Develop a model to describe the scale and proportion of atoms and molecules. Emphasize developing atomic models of elements and their numbers of protons, neutrons, and electrons, as well as models of simple molecules.
Bill Nye Cuts Cheese
Activity
INSTRUCTIONS:
1. Decide on Team Roles
2. Facilitator, read the Team Norms to your team
3. Resource Manager get the needed materials for this lab from the demo table and hand them out to your team members.
4. Team Captain will read the directions.
Directions for Activity:
A. Title the next left page in your science journal "How big is an atom?" Record the title in your table of contents along with the page number.
B. Glue the red piece of paper on this science journal page
C. Write down in your science journal, a prediction on how many times you think you will be able to cut the white piece of paper in half. You will be cutting the paper until you cannot cut a piece in half.
D. Cut the white sheet of paper in half.
E. Take one half of this paper and put it to the side. Cut the other one in half again. (Make all cuts perpendicular to the first.)
F. Repeat this process, until you can no longer cut the paper in half.
G. Record the number of cuts you were able to make in your science journal.
H. Guess how many cuts it would take to obtain a piece of paper as small as an atom. Write your guess in your science journal.
I. Together as a team, read the the document entitled "What is an Atom?"
J. Follow the directions for Experiment 2
K. Answer all of the questions from "What is an Atom?" in your science journal. Work through all questions together as a team. Discuss the answers together before deciding on an answer to write in your science journal.
| What is an Atom? | |
| File Size: | 1252 kb |
| File Type: | |
Tuesday, 3 September
HISTORY OF THE ATOM - REQUIRED
Whole Class
PURPOSE: To learn what scientists went through to understand the atom
STANDARD: 8.1.1 : Develop a model to describe the scale and proportion of atoms and molecules. Emphasize developing atomic models of elements and their numbers of protons, neutrons, and electrons, as well as models of simple molecules.
Whole Class
PURPOSE: To learn what scientists went through to understand the atom
STANDARD: 8.1.1 : Develop a model to describe the scale and proportion of atoms and molecules. Emphasize developing atomic models of elements and their numbers of protons, neutrons, and electrons, as well as models of simple molecules.
|
"History of the Atom" - Boseman Science
Watch this video (click on title above) and take notes, using the Cornell Notes Template on the right. Together as a class we will watch the documentary "Into The Atom" to help us understand what scientists know today about the atom.
INSTRUCTIONS: 1. Cut out the Pictures with the names of each of the scientists that we have been learning about. 2. You will be pasting a picture of the scientist in your science journal along with writing the information that you have collected from the previous video we watched last week and the information that you collect from watching the video "History of the Atom." Be sure that you include:
PLUS, The notes you take will help you develop your answer and the evidence to back up your answer to our phenomenon question, "Are all atoms the same?" EXTENSION ACTIVITY (Required by ALL HonOR STUDENTS)LEARNING OBJECTIVE:
Students will learn more about how scientists discovered the atom. PURPOSE: To deepen student understanding of the atom. INSTRUCTIONS: 1. Students will watch the "Into the Atom" video below. It is almost one hour long. You are welcome to watch it in smaller segments if needed. 2. Students will take Cornell Notes of the video. 3. Students will use their notes to create a timeline of the discovery of the atom.
|
| ||||||||||||
WedneSDAY, 4 September
Homework for the week: EdPuzzle #2 "Just How Small is an ATom?"
DUE: Monday, 9 September
OBJECTIVES:
1. Open the file below in Word
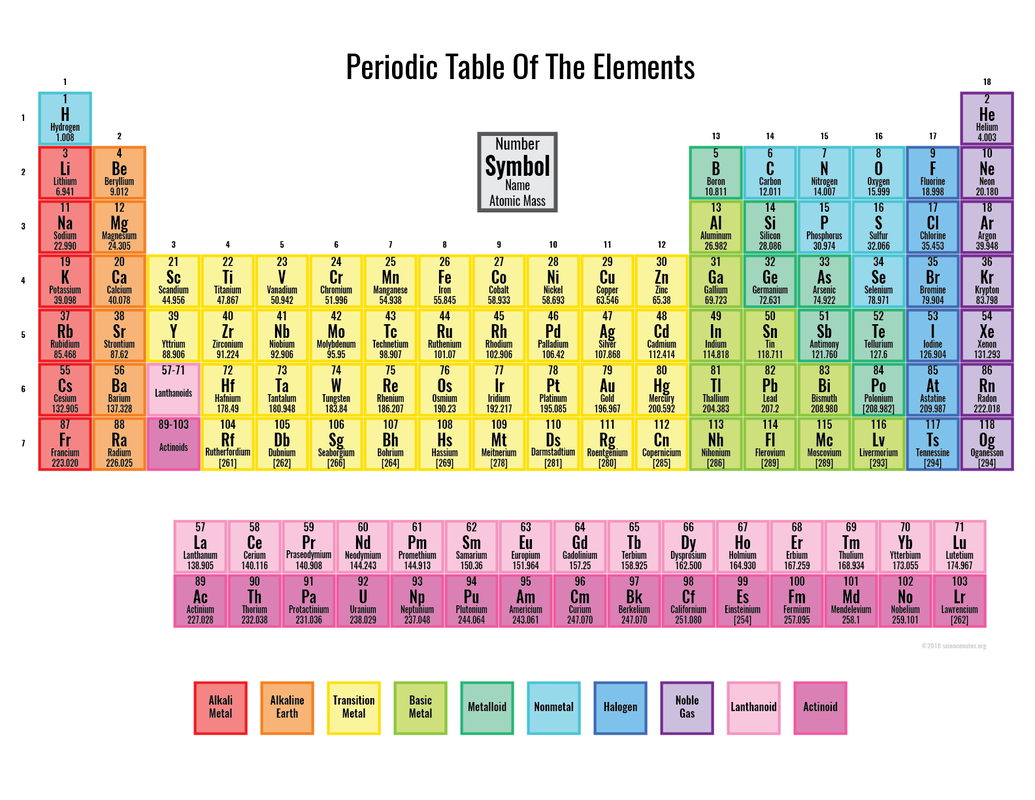
2. We will go over the parts of the periodic table together.
3. Using the information you have learned about Protons, Neutrons, and Electrons, complete the table on page two.
- Students will continue to learn about the major parts of an atom
- Students will be introduced to the Periodic Table
- Students will learn about the relationship between atoms and the Periodic Table
- Students will learn to draw Bohr models of different elements
- Complete "Introduction to Atoms and the Periodic Table" sheet
- Draw Bohr models of the first 20 elements in your science journal
1. Open the file below in Word
2. We will go over the parts of the periodic table together.
3. Using the information you have learned about Protons, Neutrons, and Electrons, complete the table on page two.
| intro_to_atoms_and_periodic_table.docx | |
| File Size: | 282 kb |
| File Type: | docx |
thursday, 5 September CLASS MEETS IN B-23
OBJECTIVES:
- Students will continue to learn about atoms and elements
- Students will build a basic 3D model (not to scale) of an atom
- Students will play the elements game using the periodic table of elements
- Students will create a basic 3D model (not to scale) of an atom
| building_atoms_instructions.pdf | |
| File Size: | 1310 kb |
| File Type: | |
Friday, 6 September
OBJECTIVES:
Students will learn how to build a MindMap
ASSIGNMENTS:
Students will build a MindMap about what they have learned about atoms
INSTRUCTIONS:
1. In the middle of a blank sheet of paper, write down our phenomenon question: "Are All Atoms the Same?"
2. Now make one large line coming out from the center question that says: Atomic Theory
3. Now make another large line coming out from the center question that says: Parts of an Atom
4. Now make another large line coming out from the center question that says: Atoms make...
5. With the "Parts of an Atom" line add a small branch for each of the parts of an atom
6. With each branch from the Parts of Atom, add smaller twigs to each branch with words that describe each part.
7. With the "Atomic Theory" line add a small branch for each scientist we have learned about - you can also add pictures if it is easier to show what you know
8. With each branch, add words on twigs that describe what you know about that scientist - you can also add pictures if it is easier to show what you know
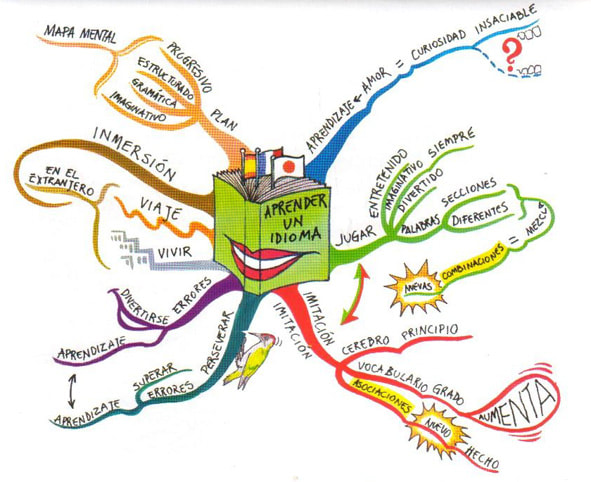
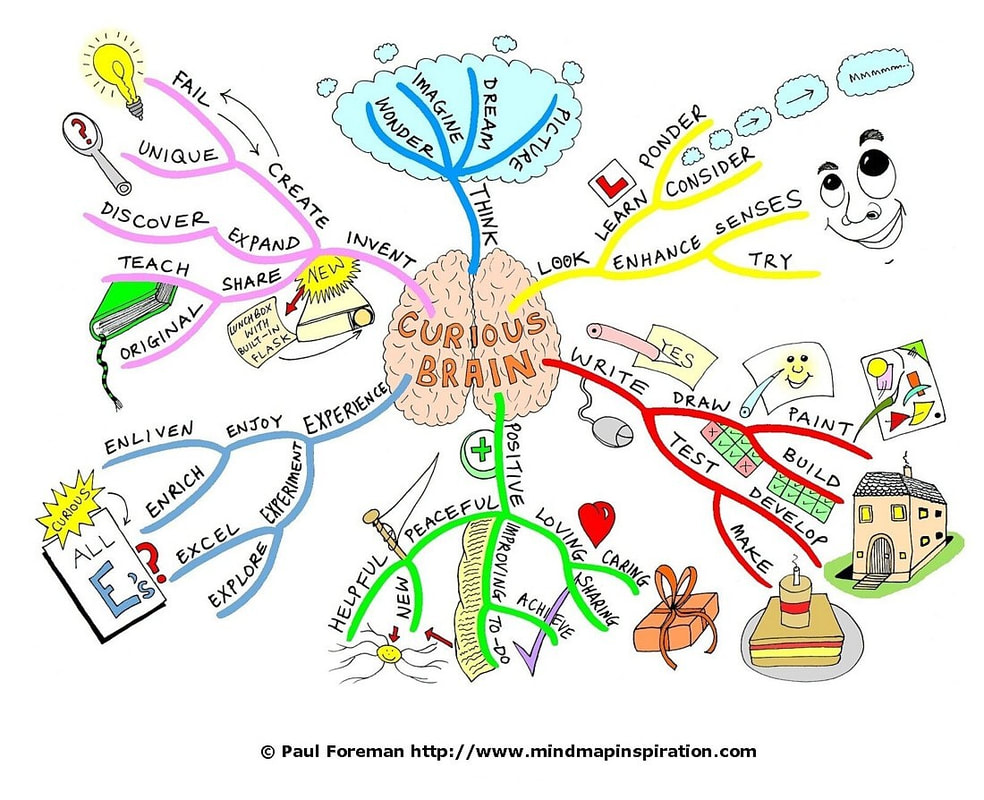
SEE EXAMPLES BELOW
Students will learn how to build a MindMap
ASSIGNMENTS:
Students will build a MindMap about what they have learned about atoms
INSTRUCTIONS:
1. In the middle of a blank sheet of paper, write down our phenomenon question: "Are All Atoms the Same?"
2. Now make one large line coming out from the center question that says: Atomic Theory
3. Now make another large line coming out from the center question that says: Parts of an Atom
4. Now make another large line coming out from the center question that says: Atoms make...
5. With the "Parts of an Atom" line add a small branch for each of the parts of an atom
6. With each branch from the Parts of Atom, add smaller twigs to each branch with words that describe each part.
7. With the "Atomic Theory" line add a small branch for each scientist we have learned about - you can also add pictures if it is easier to show what you know
8. With each branch, add words on twigs that describe what you know about that scientist - you can also add pictures if it is easier to show what you know
SEE EXAMPLES BELOW
MINDMAP - REQUIRED
Individual Work
Individual Work
MONDAY, 9 September
OBJECTIVES:
- Students will build human atoms
- Students will learn more information about the particles found in an atom
- Students will participate in creating human atoms
- Students will create a foldable in their science journal with information about protons, neutrons, and electrons
| Parts of Atoms - Foldable | |
| File Size: | 378 kb |
| File Type: | |
Tuesday, 10 September
OBJECTIVES:
- Students will build a scale version of an atom
- Students will learn that atoms combine to make molecules
- Team-built scale versions of atoms
- Team-built molecules
Some information we need to know:
- Nucleus is 10,000 times smaller than the atom AND electrons are 10,000 times smaller than the nucleus
- 99.99% of an atom's volume is empty space
- If protons and neutrons were a centimeter in diameter, then electrons would be equal to the diameter of a hair. Which would make the diameter of the entire atom greater than the length of 30 football fields
Wednesday, 11 September
OBJECTIVES:
1. Students individually completed the 3-paged "Building Molecules Worksheet" You will need colored pencils to complete the task.
- Students will learn how to build molecules.
- Students will learn about two types of Molecules - Elements and Compounds
- Building Molecules Worksheet
1. Students individually completed the 3-paged "Building Molecules Worksheet" You will need colored pencils to complete the task.
| Building Molecules Worksheet | |
| File Size: | 29 kb |
| File Type: | doc |
Thursday, 12 September
OBJECTIVES:
Students will review the questions that they created at the start of the phenomenon and share their questions and answers with their team members
2. Teams will listen carefully to their team members Questions/Answers and as a team they will determine the answer, with evidence to the phenomenon question "Are All Atoms the Same?"
3. Teams will create a poster depicting their answers (drawings are better than lots of words)
4. Teams will present their posters - class will discuss each poster
5. Students will individually complete Vocab Cards for the the following words: atom, atomic theory, neutron, proton, electron, nucleus, molecule, element, compound
https://awesomescientists.weebly.com/resources.html
6. Students will paste their Vocab Cards inside of their journals.
7. Students will review their "Are All Atoms the Same?" rubric in order to prepare for tomorrow's FINAL ANSWER
- Students will review vocabulary words associated with answering the question: "Are All Atoms the Same?"
- Students will examine their understanding to the science answer for "Are All Atoms the Same?"
- Students will work with team members to answer the question: "Are All Atoms the Same?"
- Students will complete vocal cards and glue them in their science journals
- Students will complete and present a team Final Answer Poster
- Students will check to see if they can answer their Initial Questions
Students will review the questions that they created at the start of the phenomenon and share their questions and answers with their team members
2. Teams will listen carefully to their team members Questions/Answers and as a team they will determine the answer, with evidence to the phenomenon question "Are All Atoms the Same?"
3. Teams will create a poster depicting their answers (drawings are better than lots of words)
4. Teams will present their posters - class will discuss each poster
5. Students will individually complete Vocab Cards for the the following words: atom, atomic theory, neutron, proton, electron, nucleus, molecule, element, compound
https://awesomescientists.weebly.com/resources.html
6. Students will paste their Vocab Cards inside of their journals.
7. Students will review their "Are All Atoms the Same?" rubric in order to prepare for tomorrow's FINAL ANSWER
Friday, 13 February
OBJECTIVE:
1. The Final Answer is considered a test. Students will write an essay to explain their understanding and evidence for the science question: "Are All Atoms the Same?"
2. Once their answer is complete, students will complete the rubric for the phenomenon. They will circle the corresponding level of understanding for each of the required pieces of information.
3. Students will complete the Self-Assessment Form for the phenomenon.
4. The Final Answer, the Rubric, and the Self-Assessment Form will be stapled together and turned in to Mrs. Smith
- Students will demonstrate their understanding of the phenomenon question "Are All Atoms the Same?"
- Students will evaluate their efforts and knowledge during the phenomenon "Are All Atoms the Same?"
- Students will complete a Final Answer for the question, "Are All Atoms the Same?"
- Students will complete the Rubric for "Are All Atoms the Same?"
- Students will complete the Self-Assessment for "Are All Atoms the Same?"
1. The Final Answer is considered a test. Students will write an essay to explain their understanding and evidence for the science question: "Are All Atoms the Same?"
2. Once their answer is complete, students will complete the rubric for the phenomenon. They will circle the corresponding level of understanding for each of the required pieces of information.
3. Students will complete the Self-Assessment Form for the phenomenon.
4. The Final Answer, the Rubric, and the Self-Assessment Form will be stapled together and turned in to Mrs. Smith
Final Answer
For the Final Answer, your test for this phenomenon, you will need to be in the classroom to complete this assessment. You have a choice of either writing it out on paper (provided by Mrs. Smith) OR doing it on CANVAS. Your choice. I know some of you like to use paper and pencil, while others would rather type out your answer. If you are absent, because this is a test, you will need to set up a time to come in and make the assessment up in the classroom.
RUBRIC
Please circle the level of understanding you have for each of the pieces of required knowledge.
| are_all_atoms_the_same__-_sheet1.pdf | |
| File Size: | 49 kb |
| File Type: | |
SELF ASSESSMENT
Please answer each of the questions on the assessment based on your level of effort, the evidence that you would liked considered to prove you have understood the required science concepts.
| studentselfreportingform.pdf | |
| File Size: | 37 kb |
| File Type: | |