Student Learning Goal: Standard 8.1.3
Plan and conduct an investigation and then analyze and interpret the data to identify patterns in changes in a substance's properties to determine if a chemical reaction has occurred.
22 January 2020
Student Learning Objective:
Students will be introduced to the new Phenomenon "How do Fireworks work?" incorporating the understanding of chemical reactions
INITAL ANSWER
In your science journal, start a new right-hand page with the phenomenon question, "How do Fireworks work?" as the title. Write your theory as to how fireworks work. Think about what you know and include this in your answer.
Initial Answer Poster
Each member of the team shares what they have written with the other members of their team. Once everyone has shared, decide together on what your group thinks the answer might be. Each person then uses one color of marker or colored pencil to draw something for the poster.
The poster should include:
The poster should include:
- The phenomenon question
- Drawings, words and phrases that would help to answer the question
Student Phenomenon Goal Sheet
Students will fill out their goal sheet for this phenomenon and staple or tape the top of it inside the next left hand page of their science journal. See document below.
| 8th_grade_fireworks_phenomenon.docx | |
| File Size: | 25 kb |
| File Type: | docx |
23-28 January 2020
Student Learning Objective:
Students will learn about the various signs that indicate a chemical reaction has occurred.
Signs of a Chemical Reaction Foldable
|
We will be watching a series of videos that will help you define each of the signs that a chemical reaction has occurred. You will write your definition directly under the flap. On the underside of the flap you will draw a picture of an example of this type of reaction.
The Videos are from "The Kid Should See This" site: www.thekidshouldseethis.com
|
INSTRUCTIONS:
1. Fold the ends of the blank paper in until both sides touch each other to create two flaps as seen in the photo on the left 2. Cut each flap into 5 windows, being careful to only cut to the fold line as shown in the photo on the left. 3. Label the Left side flaps as follows, in order from top to bottom: "How do I know a chemical reaction has occurred?" "Chemical Reaction Equation" "Color Change" "Flammability" "Odor/Taste" 4. Label the right side flaps as follows, in order from top to bottom: "Volume/Density Change" "Change in Matter" "Gas Bubbles" "Temperature Change" "Production of Light" |
NEXT STEPS:
1. Draw a picture of the specific type of chemical reaction on the inside flap for each of the different signs of chemical reactions.
2. Use the following sources to help you find a science description for each of the different signs of a chemical reaction - write the descriptions (in pencil) under the flap.
Sources:
1. Draw a picture of the specific type of chemical reaction on the inside flap for each of the different signs of chemical reactions.
2. Use the following sources to help you find a science description for each of the different signs of a chemical reaction - write the descriptions (in pencil) under the flap.
Sources:
- https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Fundamentals/Chemical_Change_vs._Physical_Change
- https://sciencing.com/seven-things-indicate-chemical-change-occurring-12107532.html
- https://www.classzone.com/vpg_ebooks/ml_sci_gr8/accessibility/ml_sci_gr8/page_155.pdf
- https://www.britannica.com/science/chemical-reaction/The-conservation-of-matter
- http://www.chem4kids.com/files/react_intro.html
- go back and look at the videos we watch on www.thekidshouldseethis.com - search up "Chemical Reactions"
28 February 2020
Student Learning objective:
Students will learn about chemical reactions with color change, Electrical Currents, and temperature.
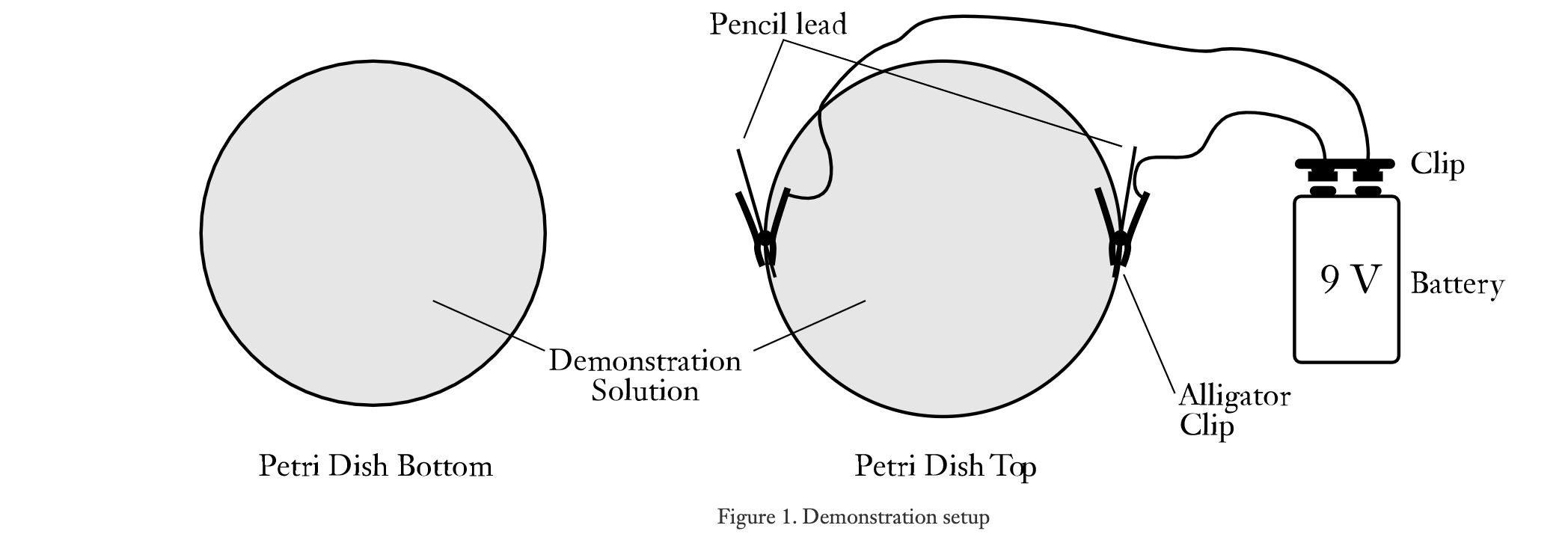
Colorful Electrolysis Lab
Materials:
Materials:
- Sodium sulfate solution
- Universal indicator solution
- Distilled water
- Battery clip with alligator ends
- 9-V Battery
- Beaker
- Graduated cylinder 10 mL
- Graduated cylinder 50 mL
- Pencil lead
- Petry dish
- Stirring Rod
|
| ||||||||||||
Safety Precautions
- Students MUST wear safety goggles, lab coat, and gloves at all times during the lab
- Do not use the Universal Indicator Solution near an open flame
- Wash hands thoroughly with soap and water before leaving the lab
Job Assignments:
Materials Manager - will be in charge of getting materials, cleaning materials, and returning materials
Facilitator - Read through the entire lab with team members before starting the lab, including the Safety Sheets; also in charge of wiping down the table during clean up.
Recorder/Reporter - Ask Mrs. Smith any clarifying questions on behalf of your team; you are responsible for making sure that all team members complete the lab sheets and turn them into Mrs. Smith. This person is also responsible for making sure the floor space around and under your table is clean during clean up.
DECIDE WHO WILL DO EACH JOB PRIOR TO STARTING TO READ THE INSTRUCTIONS
Instructions:
1. Students must read through all of the instructions before starting the lab. It is your team's responsibility to understand the lab before doing the lab. If you have any questions, have the Recorder/Reporter ask Mrs. Smith your questions. Students will take turns doing each step of the lab. Decide who will do each step prior to starting the lab.
2. Materials Manager gets materials from Mrs. Smith - you will be asked a question that you must answer correctly about the lab in order to pick up your materials.
3. LEAVE ALL MATERIALS ON THE TRAY - Pour 10 mL universal indicator solution (in 10 mL graduated cylinder) into beaker
4. Pour 40 mL sodium sulfate solution (in 50 mL graduated cylinder) into beaker and stir mixture
5. Open up petri dish and lay two halves on table.
6. Pour enough of your solution (in beaker) to cover the bottom of each half of the petri dish.
7. CAREFULLY break pencil lead in half.
8. Attach 1/2 of the lead with the alligator clips to one side of one of the petri dishes and the remaining lead to the other side of the same petri dish with the other alligator clip. The petri dishes should look like the illustration below.
Materials Manager - will be in charge of getting materials, cleaning materials, and returning materials
Facilitator - Read through the entire lab with team members before starting the lab, including the Safety Sheets; also in charge of wiping down the table during clean up.
Recorder/Reporter - Ask Mrs. Smith any clarifying questions on behalf of your team; you are responsible for making sure that all team members complete the lab sheets and turn them into Mrs. Smith. This person is also responsible for making sure the floor space around and under your table is clean during clean up.
DECIDE WHO WILL DO EACH JOB PRIOR TO STARTING TO READ THE INSTRUCTIONS
Instructions:
1. Students must read through all of the instructions before starting the lab. It is your team's responsibility to understand the lab before doing the lab. If you have any questions, have the Recorder/Reporter ask Mrs. Smith your questions. Students will take turns doing each step of the lab. Decide who will do each step prior to starting the lab.
2. Materials Manager gets materials from Mrs. Smith - you will be asked a question that you must answer correctly about the lab in order to pick up your materials.
3. LEAVE ALL MATERIALS ON THE TRAY - Pour 10 mL universal indicator solution (in 10 mL graduated cylinder) into beaker
4. Pour 40 mL sodium sulfate solution (in 50 mL graduated cylinder) into beaker and stir mixture
5. Open up petri dish and lay two halves on table.
6. Pour enough of your solution (in beaker) to cover the bottom of each half of the petri dish.
7. CAREFULLY break pencil lead in half.
8. Attach 1/2 of the lead with the alligator clips to one side of one of the petri dishes and the remaining lead to the other side of the same petri dish with the other alligator clip. The petri dishes should look like the illustration below.
9. Clip the alligators clips to the 9V battery. Be sure that each lead is submerged into the solution in the petri dish.
10. Every 2 minutes write down what you see and draw what you see on the observation lab sheet in your lab packet. You will do this 5 times during the lab.
11. While you are waiting, fill out the Energy and Matter diagram in your lab packet.
12. When the lab is determined completed, the materials manager will
14. Recorder/Reporter check floor for trash and throw away any trash found
15. Recorder/Reporter, work with your team members to complete all parts of the lab packet. Everyone completes their own packet - but remember that your team isn't done until everyone is done.
16. Recorder/Reporter turns in every members packet to Mrs. Smith at the end of class.
10. Every 2 minutes write down what you see and draw what you see on the observation lab sheet in your lab packet. You will do this 5 times during the lab.
11. While you are waiting, fill out the Energy and Matter diagram in your lab packet.
12. When the lab is determined completed, the materials manager will
- unplug the alligator clips from the battery
- unplug the alligator clips from the lead
- take the tray back to counter
- wash out the petri dishes and lay them face down on paper towels
- wash out the beaker and lay it upside down on paper towels
- wash out both graduated cylinders and place upside down on paper towels
- return the alligator clips and battery to the appropriate tray
- wash off tray, dry it, and stack it in the appropriate tray stack
14. Recorder/Reporter check floor for trash and throw away any trash found
15. Recorder/Reporter, work with your team members to complete all parts of the lab packet. Everyone completes their own packet - but remember that your team isn't done until everyone is done.
16. Recorder/Reporter turns in every members packet to Mrs. Smith at the end of class.
29 January 2020
Student Learning Objective:
Discussion/Explanation:
1. Before reviewing the scientific explanation look up the following science words and write them in your science journal (HINT: When looking up words be sure to search for the science definition of the word, example: "science definition of anode")
When an electric current is passed through an aqueous solution containing an electrolyte (Na2SO4), the water molecules break apart or decompose into their constituent elements, hydrogen and oxygen. The overall reaction occurs as two separate, independent half-reactions. Reduction of the hydrogen atoms to elemental hydrogen (H2) occurs at the cathode (–) while oxidation of the oxygen atoms in water to elemental oxygen (O2) occurs at the anode (+). Each half-reaction is accompanied by the production of OH– or H+ ions as shown:
Cathode: 4e– + 4H2O → 2H2(g) + 4OH–
Anode: 2H2O → O2(g) + 4H+ + 4e–
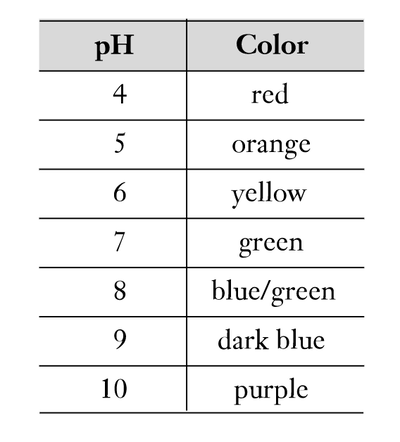
At the cathode the excess OH– will cause the pH to increase, resulting in a color change of the universal indicator solution from green (neutral, pH 7) to purple (basic, pH ≥ 10).
At the anode the excess H+ will cause the pH to decrease, resulting in a color change of the universal indicator solution from green to an orange/red color (acidic, pH ≤ 4). The electrolysis half-reactions can also be followed by observing the production of gas bubbles at the cathode (H2) and anode (O2).
Universal indicator is an acid–base indicator that is different colors at different pH values. All colors will be visible in the Petri dish as electrolysis progresses and as the pH conditions continually change due to diffusion and neutralization.
3. Together as a team, work on completing the Discussion Questions found in your Lab Packet.
1. Before reviewing the scientific explanation look up the following science words and write them in your science journal (HINT: When looking up words be sure to search for the science definition of the word, example: "science definition of anode")
- Anode
- Cathode
- Electrolysis
- Basic (pH base)
- Acidic (pH acid)
When an electric current is passed through an aqueous solution containing an electrolyte (Na2SO4), the water molecules break apart or decompose into their constituent elements, hydrogen and oxygen. The overall reaction occurs as two separate, independent half-reactions. Reduction of the hydrogen atoms to elemental hydrogen (H2) occurs at the cathode (–) while oxidation of the oxygen atoms in water to elemental oxygen (O2) occurs at the anode (+). Each half-reaction is accompanied by the production of OH– or H+ ions as shown:
Cathode: 4e– + 4H2O → 2H2(g) + 4OH–
Anode: 2H2O → O2(g) + 4H+ + 4e–
At the cathode the excess OH– will cause the pH to increase, resulting in a color change of the universal indicator solution from green (neutral, pH 7) to purple (basic, pH ≥ 10).
At the anode the excess H+ will cause the pH to decrease, resulting in a color change of the universal indicator solution from green to an orange/red color (acidic, pH ≤ 4). The electrolysis half-reactions can also be followed by observing the production of gas bubbles at the cathode (H2) and anode (O2).
Universal indicator is an acid–base indicator that is different colors at different pH values. All colors will be visible in the Petri dish as electrolysis progresses and as the pH conditions continually change due to diffusion and neutralization.
3. Together as a team, work on completing the Discussion Questions found in your Lab Packet.
Class Discussion:
1. After teams have completed the Discussion Questions, the class will go over the answers together. Recorder/Reporter please be ready to report what your team came up with as an answer to any of the questions.
2. Students work on the Cause and Effect sheet together with their team.
3. At this point, the entire lab packet should be completed. Facilitator please collect your team's packets and turn them into Mrs. Smith.
1. After teams have completed the Discussion Questions, the class will go over the answers together. Recorder/Reporter please be ready to report what your team came up with as an answer to any of the questions.
2. Students work on the Cause and Effect sheet together with their team.
3. At this point, the entire lab packet should be completed. Facilitator please collect your team's packets and turn them into Mrs. Smith.
30 January 2020
Student Learning Objectives:
Students learn about concepts involving chemical reactions
INSTRUCTIONS:
1. Students will pick three of the following articles to learn more about how chemical reactions works and other concepts that tie to chemical reactions.
pH "What are acids and bases?"
"Periodic Table"
"How water is cleaned up for drinking"
"...Firenados"
"Changing the world with chemistry"
"Sublimination"
"...Hydrogen into metals"
"What is a catalyst?"
"Scientists know you pee in the pool"
"Photochromic"
"Recycling urine..."
"Why onions make us cry"
"Bitter + Bitter = Sweet"
"Moisture unmasked hidden message"
"Luminescence"
"Knowing what you don't know can help..."
2. Read the article and determine whether or not there is a chemical reaction involved.
3. If there is a chemical reaction involved, then determine what causes the chemical reaction and how do we know there is a chemical reaction occurring (HINT: Use your foldable to help you determine how we know there is a chemical reaction occurring).
4. On a piece of paper you will write: Title of the article, what the chemical reaction is, what caused it, and how we would know this chemical reaction is happening. You will do this for all three articles you pick. You will also need to pick 3 of the POWER WORDS that you think would help you learn more about chemical reactions. Write the words only on the paper.
5. Next, on a vocab square, write each of the POWER Words you have chosen, along with its definition (provided at the end of the article), then finish filling out the Vocab Square. Cut the Vocab Squares out and paste them in your science journal.
6. Turn in your paper with your new found knowledge of chemical reactions that occur around us.
1. Students will pick three of the following articles to learn more about how chemical reactions works and other concepts that tie to chemical reactions.
pH "What are acids and bases?"
"Periodic Table"
"How water is cleaned up for drinking"
"...Firenados"
"Changing the world with chemistry"
"Sublimination"
"...Hydrogen into metals"
"What is a catalyst?"
"Scientists know you pee in the pool"
"Photochromic"
"Recycling urine..."
"Why onions make us cry"
"Bitter + Bitter = Sweet"
"Moisture unmasked hidden message"
"Luminescence"
"Knowing what you don't know can help..."
2. Read the article and determine whether or not there is a chemical reaction involved.
3. If there is a chemical reaction involved, then determine what causes the chemical reaction and how do we know there is a chemical reaction occurring (HINT: Use your foldable to help you determine how we know there is a chemical reaction occurring).
4. On a piece of paper you will write: Title of the article, what the chemical reaction is, what caused it, and how we would know this chemical reaction is happening. You will do this for all three articles you pick. You will also need to pick 3 of the POWER WORDS that you think would help you learn more about chemical reactions. Write the words only on the paper.
5. Next, on a vocab square, write each of the POWER Words you have chosen, along with its definition (provided at the end of the article), then finish filling out the Vocab Square. Cut the Vocab Squares out and paste them in your science journal.
6. Turn in your paper with your new found knowledge of chemical reactions that occur around us.
31 January 2020
Student Learning Objectives:
Students will review the basic concepts involving chemical reactions
Max Axiom and Chemical Reactions
Students will read the graphic novel "Max Axiom and Chemical Reactions" individually while answering questions. Once everyone on the team has finished reading and answering questions, students will compare answers and discuss any answers that differ with one another.
6 February 2020
Student Learning Objectives:
Students will review/learn about atoms, the periodic table, chemical reactions, Compounds and Mixtures, as well as the Law of conservation of matter - all having to do with how fireworks work.

BrainPop Assignments:
Carefully go through each video and complete the assigned tasks that go with each video.
To log in: You will need our class code: lasso7975 and your log in for BrainPop which should be: User: your 9 number Password: westlake1
If you have any issues let Mrs. Smith know and we'll get things fixed. Click below to go to the various assignments! Enjoy!
Video#1:www.brainpop.com/dashboard/assignments/d4a3b61732f1a73b/periods/4240fe869826ca16
Video #2: https://www.brainpop.com/dashboard/assignments/48e0f9c0bd917ace/periods/4240fe869826ca16
Video #3: https://www.brainpop.com/dashboard/assignments/eaae0edd7a65cfef/periods/4240fe869826ca16
Video #4: https://www.brainpop.com/dashboard/assignments/f64aab39144e79a3/periods/4240fe869826ca16
Video #5: https://www.brainpop.com/dashboard/assignments/09dfc30263c62239/periods/4240fe869826ca16
Science Journal Assignment:
Once you have watched all of the videos and completed all of the assigned tasks. In your science journal write down how each of the topics: Atoms, the Periodic Table, Chemical Reaction Equations, Compounds and Mixture, and the Law of Conservation of Matter have to do with Fireworks.
Carefully go through each video and complete the assigned tasks that go with each video.
To log in: You will need our class code: lasso7975 and your log in for BrainPop which should be: User: your 9 number Password: westlake1
If you have any issues let Mrs. Smith know and we'll get things fixed. Click below to go to the various assignments! Enjoy!
Video#1:www.brainpop.com/dashboard/assignments/d4a3b61732f1a73b/periods/4240fe869826ca16
Video #2: https://www.brainpop.com/dashboard/assignments/48e0f9c0bd917ace/periods/4240fe869826ca16
Video #3: https://www.brainpop.com/dashboard/assignments/eaae0edd7a65cfef/periods/4240fe869826ca16
Video #4: https://www.brainpop.com/dashboard/assignments/f64aab39144e79a3/periods/4240fe869826ca16
Video #5: https://www.brainpop.com/dashboard/assignments/09dfc30263c62239/periods/4240fe869826ca16
Science Journal Assignment:
Once you have watched all of the videos and completed all of the assigned tasks. In your science journal write down how each of the topics: Atoms, the Periodic Table, Chemical Reaction Equations, Compounds and Mixture, and the Law of Conservation of Matter have to do with Fireworks.
18 February 2020
Student Learning objective:
Students will learn about the basics of how fireworks work
Students will learn about how fireworks and chemical reactions are connected

Materials Needed:
Students will go through a list of statements having to do with fireworks and mark down whether or not they think the statement is correct. (See Fireworks Lesson 1 Worksheet) From there, students will read through the ChemMatters article, "Fireworks" and find out what the text says about the same statements.
TO DO:
***** BE YOUR BEST STUDENT TODAY,
I'M COUNTING ON YOU*****
1. Write the Student Learning Objectives in your planner
2. Answer the first 4 questions (pg 2),
3. Then fill out the "Me" column of the Fireworks Lesson 1 Worksheet (pg 3-4)
4. Carefully read the ChemMatters article "Fireworks." Look for information in the article that matches up with or disagrees with the statements from the worksheet.
5. Now go back to the Fireworks Lesson 1 Worksheet and find out what the article had to say about the statements on the worksheet. In the space under each statement, write a statement from the article that supports or refutes(disagrees) with the original statement.
6. Turn your worksheet into the teacher
- ChemMatters Article "FireWorks" one for each student
- Pencil
- "Fireworks" worksheet #1
Students will go through a list of statements having to do with fireworks and mark down whether or not they think the statement is correct. (See Fireworks Lesson 1 Worksheet) From there, students will read through the ChemMatters article, "Fireworks" and find out what the text says about the same statements.
TO DO:
***** BE YOUR BEST STUDENT TODAY,
I'M COUNTING ON YOU*****
1. Write the Student Learning Objectives in your planner
2. Answer the first 4 questions (pg 2),
3. Then fill out the "Me" column of the Fireworks Lesson 1 Worksheet (pg 3-4)
4. Carefully read the ChemMatters article "Fireworks." Look for information in the article that matches up with or disagrees with the statements from the worksheet.
5. Now go back to the Fireworks Lesson 1 Worksheet and find out what the article had to say about the statements on the worksheet. In the space under each statement, write a statement from the article that supports or refutes(disagrees) with the original statement.
6. Turn your worksheet into the teacher
On the plane - took 2 days to get to Méribel, France due to plane problems, but we made it!
The French Alps
Ready to Ski!
|
View from the condo in Méribel/Morel (above)
My husband and our son Benone (below) I hope you all had a fun 4-day weekend. We spent almost all of our traveling here to Méribel. Wish you were all here with us. It's so beautiful. Believe it or not the altitude here is lower than when you're in the Wasatch mountains.
Be your BEST! | ||||||||||||
19 February 2020
Student Learning Objectives:
Students will continue to learn about how fireworks are created and work

Materials Needed:
Students will reread the ChemMatters article "Fireworks" and complete the Fireworks Lesson #2 worksheet
TO DO:
WORK ON YOUR FUTURE TODAY BY BEING YOUR BEST STUDENT - YOUR FUTURE SELF IS COUNTING ON IT!
1. Write your Student Learning Objective in your planner.
2. Reread the article "Fireworks," while reading complete the chart below describing how fireworks work, and whatpyrotechnic chemists do.
3. On the last page of the worksheet, find references to the various chemical named in the article. Write their formulas, names and uses in the table.
4. Turn in your worksheet to the teacher.
- ChemMatters Article "FireWorks" one for each student
- Pencil
- "Fireworks" worksheet #2
Students will reread the ChemMatters article "Fireworks" and complete the Fireworks Lesson #2 worksheet
TO DO:
WORK ON YOUR FUTURE TODAY BY BEING YOUR BEST STUDENT - YOUR FUTURE SELF IS COUNTING ON IT!
1. Write your Student Learning Objective in your planner.
2. Reread the article "Fireworks," while reading complete the chart below describing how fireworks work, and whatpyrotechnic chemists do.
3. On the last page of the worksheet, find references to the various chemical named in the article. Write their formulas, names and uses in the table.
4. Turn in your worksheet to the teacher.
|
|
| ||||||||||||||||||
Today, Tuesday, we opted to take a 2-hour drive through the Alps to Geneva, Switzerland. It rained a lot and snowed a little making skiing conditions not so good, so a day in Geneva sounded like just the thing to do.
The first photo is what we woke up to this morning and the one under it is of Mont Blanc. There's a photo of my bad day of skiing, and a photo of Ben pointing to the condo where we are staying. The other photos we took today in Geneva. We climbed to the towers of Saint Pierre Eglise (church) today, and got some great shots of the city.
As you can see Chip and Ben got to eat fondue, however, my trout was outstanding, yummy.
Wish you were here!
Hope you're doing your best and being your best selves today. :)
The first photo is what we woke up to this morning and the one under it is of Mont Blanc. There's a photo of my bad day of skiing, and a photo of Ben pointing to the condo where we are staying. The other photos we took today in Geneva. We climbed to the towers of Saint Pierre Eglise (church) today, and got some great shots of the city.
As you can see Chip and Ben got to eat fondue, however, my trout was outstanding, yummy.
Wish you were here!
Hope you're doing your best and being your best selves today. :)
20 February 2020
STudent Learning Objective:
Students will continue to learn about the connections between chemical reactions and how fireworks work
Students will continue to learn about the connections between chemical reactions and how fireworks work
Assignment Overview:
1. Put their names on the paper.
2. Search for the science definitions of the words and laws on the first side of their paper and write down their findings
3. Put away their Chromebooks.
4. Reread the article, "Fireworks"
5. Answer questions on the worksheet when they have finished re-reading the article.
6. Turn in your worksheet to the teacher.
- Students will work on understanding some key terms and laws of science by finding their meanings
- Students will reread ChemMatters article "Fireworks" and find additional answers.
1. Put their names on the paper.
2. Search for the science definitions of the words and laws on the first side of their paper and write down their findings
3. Put away their Chromebooks.
4. Reread the article, "Fireworks"
5. Answer questions on the worksheet when they have finished re-reading the article.
6. Turn in your worksheet to the teacher.
|
|
| ||||||||||||||||||